We’ve all heard about the opioid epidemic and the number of fatalities that rose for decades. We heard about the Sackler family at Purdue Pharma and the criminal activities surrounding OxyContin, as well as other bad actors that include additional large pharma companies and distributors. We’ve heard about the Fentanyl crisis and how that synthetic opioid has been a primary cause for overdoses in the past several years.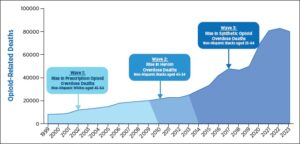
If you’ve followed the stories a bit closer, you’ve likely heard about other elements. We’re going to dive a bit deeper, though, not at the illegal drug trade, but at the system surrounding opioid addiction, treatment, and recovery.
The most common recommended course of treatment for opioid-dependent people includes opioid maintenance medication. The primary argument is that it prevents overdose fatalities and keeps people from returning back to illicit drug use. While that may be true in the short term, if it were true as a whole then the overdose fatalities shouldn’t have skyrocketed the way they did.
We seemed to have quickly dismissed the data surrounding the 15-20% of returning solders from Vietnam being addicted to heroin. Somewhere between 75% and 95% of them simply stopped when returning to life back in the U.S. This recovery was achieved without going to treatment. I’d be willing to bet that very few people in today’s treatment landscape are aware of this fact.
I believe that long-term opioid maintenance drugs create a holding pattern before people decide to get off opioids permanently. Yes, there are certainly examples where this is not the case, but having been in the addiction treatment space and worked with hundreds of clinicians and thousands of patients in recovery, I don’t make these statements casually.
Methadone was first approved by the FDA in 1947 as a painkiller, and was later beginning to be used in treating heroin addiction as early as the 1960s. Methadone maintenance grew to the introduction of Opioid Treatment Programs (OTPs) by 1969 and was officially approved for treating opioid addiction in 1972. While not producing the same euphoric effect of heroin or other painkillers, methadone is more difficult to withdraw from after becoming physically dependent.
People receiving methadone maintenance through OTPs grew to over 100,000 by the mid-90s, topping 350,000 by 2015, and today there are approximately half a million people on methadone daily.
A similar drug in this category called buprenorphine was first approved by the FDA in 1985. The Federal Government co-developed a version of the drug with Reckitt Benckiser pharmaceuticals to be approved for opioid addiction treatment in 2002. More than $62M in public funds was used to develop the drug, which was named Suboxone, and it received a patent for 7 years given its new formulation that included the opioid antagonist naloxone. The combination was supposed to prevent abuse by including naloxone in its formulation, though it was quickly found on the black market along with other painkillers and heroin. To keep the patent, and higher prices that come with it, Reckitt Benckiser introduced sublingual strips in 2010. It wasn’t until 2018 that generics finally hit the market. The company routinely generated over a billion dollars in sales annually for Suboxone.
When Suboxone was first introduced, it was primarily used as a short term stabilization and quick tapering process for easing withdrawal symptoms, but then more patients were encouraged to take it as part of a long term maintenance drug. It was also approved for office-based settings, though patient enrollment was limited initially to 30 per doctor. This increased to 100 patients per doctor by 2006, and then up to 275 patients by 2016. By 2022, the limits were removed completely.
The number of patients on buprenorphine for opioid addiction when from 12,000 in 2002 and quickly rose to 300,000 by 2006. Usage continued to rapidly escalate to roughly 1.5 million by 2023.
In 2019 the Department of Justice fined Reckitt Benckiser a total of $1.4B, and the press release stated: “Global consumer goods conglomerate Reckitt Benckiser Group plc (RB Group) has agreed to pay $1.4 billion to resolve its potential criminal and civil liability related to a federal investigation of the marketing of the opioid addiction treatment drug Suboxone. The resolution – the largest recovery by the United States in a case concerning an opioid drug – includes the forfeiture of proceeds totaling $647 million, civil settlements with the federal government and the states totaling $700 million, and an administrative resolution with the Federal Trade Commission for $50 million.
Suboxone is a drug product approved for use by recovering opioid addicts to avoid or reduce withdrawal symptoms while they undergo treatment. Suboxone and its active ingredient, buprenorphine, are powerful and addictive opioids.
“The opioid epidemic continues to be a serious crisis for our nation, and I’m proud of the work the Department of Justice and our partners are doing to address this epidemic,” said Principal Deputy Associate Attorney General Claire Murray.
“We are confronting the deadliest drug crisis in our nation’s history. Opioid withdrawal is difficult, painful, and sometimes dangerous; people struggling to overcome addiction face challenges that can often seem insurmountable,” said Assistant Attorney General Jody Hunt for the Department of Justice’s Civil Division. “Drug manufacturers marketing products to help opioid addicts are expected to do so honestly and responsibly.”
A year later, the Department of Justice issued another release regarding RB Group’s subsidiary Indivior, stating “Indivior Solutions today pleaded guilty to a one-count felony information and, together with its parent companies Indivior Inc. and Indivior plc, agreed to pay a total of $600 million to resolve criminal and civil liability associated with the marketing of the opioid-addiction-treatment drug Suboxone.”
In 2021, Indivior was fined another $300 million for fraud. The California Attorney General at the time announced the settlement against resolving claims that Indivior falsely and aggressively marketed Suboxone, resulting in improper use of state Medicaid funds.
In 2023 Indivior paid another settlement of $102.5 million to states for its illegal monopolistic practices.
Here is a perfect example of how easily people forget the role that those who were supposed to be helping people were actively perpetuating the problem. It didn’t get nearly as much coverage as other parts of the opioid epidemic. And let’s not forget that multiple government agencies aided and abetted their activities, including heavy promotion for the use of the drug and of course helping them formulate it and keep its patents long-term so that the fines were relatively insignificant to the amount of money they made.
The number of OTPs started growing so rapidly during the early 2000s that several states placed a moratorium on new licenses. Today there are about 2,200 OTPs throughout the country providing typically either methadone or some form of buprenorphine to 600,000 patients. The remainder of patients are seen by doctors in office-based settings or other outpatient treatment programs.
OTP Regulations
While each state has a set of standards for Opioid Treatment Programs, Federal law is very relaxed around enforcing anything more than the minimum standards created by the Substance Abuse and Mental Health Services Administration (SAMHSA).
There was an update to these standards in the Code of Federal Regulations in 2024.
“Many patients benefit from a full range of more intensive supports, including psychosocial services and medical treatment. Often, intensity and duration may be greater at the beginning of the treatment or at times of OUD recurrence. Since determination of services and service frequency is a shared decision between both the OTP multidisciplinary team and the patient, staff will need to skillfully navigate the therapeutic alliance to address concerns of vulnerability with the patient. Unless therapeutic benefits no longer outweigh risks, there should be no limits on a patient’s duration of treatment or dosage level of medication. Likewise, there should be no limitations on the psychosocial services offered to patients, even when they no longer receive medication. Programs should make every effort to retain patients in treatment as long as it is clinically appropriate, medically necessary, and acceptable to the patient. Maintaining a patient on medication, even when psychosocial services or other clinic services may not be yielding optimum results, is beneficial to both the individual patient and public health.”
As stated on the SAMHSA website, counseling and supportive services are supposed to be tailored to each individual’s needs, while not laying out clear minimums. “Importantly, if a patient is not interested in participating in counseling, their refusal does not preclude them from receiving MOUD.”
This is contrary to even Intensive Outpatient Services (IOP), for example, which require a minimum of 9 hours of therapeutic delivery each week, which is considered one of the lowest levels of care for addiction treatment. Even regular outpatient services provide more individual and group support than a typical OTP does.
The rationale is that if they keep people on opioids, they are statistically less likely to overdose since they’re not using as many street drugs. However, there are many other challenges.
SAMHSA recognizes on their website that these regulations, especially the updates, are broad and relaxed. It says states are allowed to set additional regulations as they choose, but stop well short of actually requiring more structured programming.
The average OTP patient on Medicaid has 1.66 treatment episodes per year, and approximately 25% have had a previous treatment episode within the past 60 days. There is a paradox of lack of treatment plan adherence coupled with long-term dependence on the medication. The average OTP patient goes on and off maintenance drugs like methadone or buprenorphine for years. The investigative report published in the Journal of the American Medical Association Network studied over 260,000 Medicaid beneficiaries in OTP programs and “found low retention rates and variable treatment duration across OTPs. This result indicates opportunities for quality improvement.”
More than 75% of the OTPs accept Medicaid, and more than half of their patients are covered by Medicaid. In California, for example, that number can be up to 80%. Therefore, Medicaid is the largest single payer for OTPs.
As I shared in an earlier episode, the last treatment center that I was personally an owner of did well with the dozens of people we served at a time in an outpatient setting. However, within just a couple miles there were three OTPs with nearly 300 patients in them each. I knew most of them were stuck in a rut and weren’t getting much help beyond a monthly counseling session, some meetings here and there, and their daily maintenance drugs.
In 2018 I vowed to myself that one day we had to find a way to help people get out of this rut that was affordable and accessible.
I was naïve to believe that most of these programs cared enough to try and help people get out of the stuck loops vs. just keeping them from going into withdrawals. Yes, the people who work there care for the individuals they treat. I mean the business models of the companies.
I started reaching out to various OTP clinics in 2022 and 2023 with ways they could provide more help to their patients in a highly affordable and accessible way. Crickets. Literally not one single email reply.
Then, I tried to reengage via professional contacts with two sets of multi-state OTP providers, each of which treat tens of thousands of patients annually. We had meetings with C-suite personnel where I presented them with multiple ways for them to be able to offer additional supportive services that would add and/or help recapture revenue for existing services while providing more help for their patients in other parts of their lives. Not surprisingly, they were hesitant.
One of them initially agreed to a pilot, then later responded saying, “based on our previous discussion around reimbursement rates and only offering group counseling, we could not make it make sense for us to move forward right now.” In other words, it wasn’t enough of a bump in revenue for them to put forth the effort, regardless of how much it would have helped their patients in other areas of their lives. They just wanted to be able to bill for individual therapy sessions at the highest allowable rates on top of the daily medications.
The other one came back and eventually said that their clinical team felt it would be an “administrative distraction” to provide additional support to patients beyond minimum requirements.
These programs print money, with relatively low oversight. In August of 2025, New Hampshire Senator Maggie Hassan of the U.S. Senate Subcommittee on Finance and Health Care initiated an investigation into the conduct, issuing letters to CEOs of at least four large OTP providers, stating, “In addition, some for-profit OTP companies appear to systematically ignore the requirements that they claim to support. OTPs owned by these companies have allegedly falsified patient mental health records and engaged in other misconduct that enabled them to fraudulently bill insurance and increase revenue.”
Senator Hassan and colleagues previously also started asking questions about the impact of Private Equity in the space, something we’ll get into more for another episode.
Hopefully this and similar investigations into the industry continues and will bear fruit for systemic change. However, without enough public scrutiny, I’m skeptical that it will end in little more than paying fines, issuing a press release, and going on with business as usual.
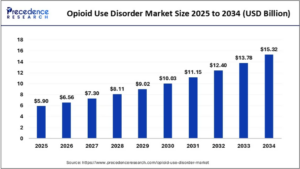 Market research predicts that the global opioid use disorder market is predicted to increase from USD 6.56 billion in 2026 to approximately USD 15.32 billion by 2034. The increasing rates of substance use disorders are the key factor driving the market growth. Also, rising government efforts to deal with these disorders, coupled with the increasing awareness of opioid use conditions, can fuel market growth shortly. North America dominated the global opioid use disorder market with the largest share of 69% in 2024.
Market research predicts that the global opioid use disorder market is predicted to increase from USD 6.56 billion in 2026 to approximately USD 15.32 billion by 2034. The increasing rates of substance use disorders are the key factor driving the market growth. Also, rising government efforts to deal with these disorders, coupled with the increasing awareness of opioid use conditions, can fuel market growth shortly. North America dominated the global opioid use disorder market with the largest share of 69% in 2024.
However, pointing out things that aren’t working doesn’t do much good alone without some kind of solutions.
Naltrexone has been available for decades, which blocks the euphoric and sedative effects of opioids such as heroin, morphine, and codeine. Naltrexone binds and blocks opioid receptors and reduces and suppresses opioid cravings. Naltrexone is not an opioid, is not addictive, and does not cause withdrawal symptoms with stop of use.
An injectable form of naltrexone called Vivitrol was approved to treat opioid use disorder twenty years ago, and generally lasts about 30 days. One key factor for success to length of time, where repeated monthly injections are recommended, as recovery rates increase after 90 days of abstinence, and continue to improve after 6 and 12 months.
Implantable forms of naltrexone are available through certain compounding pharmacies, and the National Institute of Drug Abuse has recently provided grants for human clinical trials. When trials are completed and full FDA approval is granted for widespread use, these pellets typically last three times longer than the injectable form, therefore increasing likelihood of success for other treatment and therapeutic actions.
A study published by the NIDA in 2017 found extended release naltrexone to be just as effective post-detox as Suboxone, yet without the patients still being physically dependent on opioids.
Lofexidine (brand name Lucemyra) is the only FDA-approved non-opioid medication specifically indicated to mitigate opioid withdrawal symptoms in adults. Approved in 2018, it is used to manage symptoms like vomiting, diarrhea, and muscle aches for up to 14 days. While not a treatment for opioid use disorder (OUD) itself, it can be part of a broader, long-term management plan.
There are plenty of inpatient and outpatient detox services and protocols, including even some neurostimulation devices that have received FDA clearance for reducing the opioid withdrawal symptoms as well.
Getting people fully detoxed first and then onto some form of extended release naltrexone long enough for counseling, education, and supportive services to help produce lasting change. Medicaid rates for bundled OTP services are about $300 per patient per week (estimated at nearly $5 billion annually). Not only would costs decrease dramatically, but less people would stay on Medicaid and a higher percentage would be contributing more to society. And, more importantly, more effective treatment protocols would save more lives.